  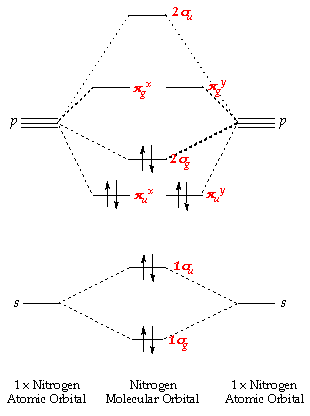
That's why the F1s orbital does not be considered here since it's full-filled.Īs mentioned before, if an atomic orbital cannot find any other orbital with similar symmetry, then it will remain as non-bonding molecular orbital. For example, s orbital can’t overlap with p x orbital if the overlap equally with both the same and opposite signs, this will cancel the bonding and anti-bonding effect which will result in no molecular orbital forms.īesides that, we normally only consider the valence atomic orbitals. Īlso notice that only two atomic orbitals with similar symmetry properties can combine together. That's why H1s will form molecular orbitals with F2p instead of F2s. Comparing with F2s orbital which has -40.17eV as its orbital potential energy, they are really close to each other. in the hydrogen fluoride HF, hydrogen's 1s orbital will overlap with one of the fluorine's 2p orbitals to form a σ orbital and a σ * orbital since the orbital potential energy of H1s orbital is -13.61eV and of F2p is -18.65eV. The 1s orbital cannot overlap with 2s orbital in this case.įor another example, in a heteronuclear diatomic molecule, which means that the atoms in the molecule are different elements, the orbitals with the closest energy can overlap with each other and form molecular orbitals.Įg. in the oxygen O 2, the 1s orbital will overlap with 1s orbital to form a σ orbital and a σ * orbital and the 2s orbital will overlap with 2s orbital to form a σ orbital and a σ * orbital. For example, in a homonuclear diatomic molecule, which means that both atoms are the same element, the same orbitals will overlap together and form molecular orbitals.Įg. Therefore, the atomic orbitals generally tend to overlap one by one from the lowest potential energy to the highest potential energy. Normally, in diatomic molecular orbitals, the atomic orbitals with the closest energy level can overlap with each other and form molecular orbitals. That’s why they are called “non-bonding” molecular orbitals. These orbitals only exist when some atomic orbitals of an atom cannot find any atomic orbital from another atom that has the same symmetry properties, then these atomic orbitals will remain at the same energy and form no bond. This bonding and anti-bonding orbitals caused by the different ways of overlapping of the atomic orbitals.īesides the bonding and anti-bonding molecular orbitals, there can also be some non-bonding orbitals. For example, if there is a 1σ bonding orbital, then there must be a 1σ *, which is the relevant anti-bonding orbital of 1σ, where * is used to represent anti-bonding. The bonding orbitals and anti-bonding orbitals always have the same number and relate with each other. In a molecular orbital diagram, if atomic orbitals form a bonding orbital, they must form an anti-bonding orbital. When two atomic orbitals overlap, they can form new orbitals in two ways: one is the bonding orbital and another one is the anti-bonding orbital. This means that when the atoms get closer to each other, their atomic orbitals can overlap and the probability of the occurring of the electrons from the atoms becomes significantly in the overlap regions, which is the formation of the molecular orbitals.īonding, Anti-bonding and Non-bonding Molecular Orbitalsīefore it goes further, some acknowledge about bonding and anti-bonding should be emphasized. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
